|
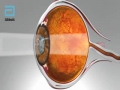
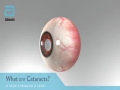
Abbott announced today that the U.S. Food and Drug Administration (FDA) has approved the Tecnis Symfony® Intraocular Lenses for the treatment of cataracts. The first in a new category of intraocular lenses (IOLs), the Tecnis Symfony lenses are the only lenses in the United States that provide a full range of continuous high-quality vision following cataract surgery, while also mitigating the effects of presbyopia by helping people focus on near objects. The FDA approval includes a version of the lens for people with astigmatism, the Tecnis Symfony Toric IOL.
Cataracts are a common condition, with almost 4 million cataract surgeries performed each year, and that number is expected to increase.1 By age 80, more than half of all Americans either have a cataract or have had cataract surgery.2 However, cataracts do not just impact seniors. In 2016 it is estimated that nearly one in four cataract surgeries will be performed on people younger than 65.1 Many people who have cataracts experience other problems with their vision, such as presbyopia and astigmatism, which the Symfony lenses also address. Presbyopia, which affects most people over age 40, means people have lost the ability to focus on objects up close and often require glasses to perform near visual tasks. Astigmatism is when the cornea is misshapen, which causes blurry or distorted vision.
To view the multimedia release go to:
http://www.multivu.com/players/English/7870151-abbott-intraocular-lenses/
Categories //
Miscellaneous
Added:
3562 days ago by
MultiVuVideos
Runtime: 0m35s | Views: 1396 |
Comments: 0
Not yet rated |
|