|
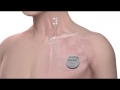
LivaNova PLC (NASDAQ:LIVN; LSE: LIVN) (“LivaNova” or the “Company”), a market-leading medical technology and innovation company, today recognizes that more than one million people in the United States with drug-resistant epilepsy continue to be treated unsuccessfully with drugs, despite the proven effectiveness of non-drug treatment options. LivaNova’s VNS Therapy®, for example, is a medical device treatment specifically designed for people with drug-resistant epilepsy that is already used by more than 85,000 people around the world.
One in three people with epilepsy has the type that is not well controlled with drugs, called drug-resistant epilepsy. For those people, prescribing more medicine is usually not the most effective option. In fact, research shows that if you haven’t found seizure freedom after your first two drugs, there is a 95 percent chance that no drug or combination of drugs will result in seizure freedom.
“Despite what we know about drug-resistant epilepsy, the majority of people affected by this condition will continue to be prescribed additional medications instead of being evaluated for surgery or seen in a specialized epilepsy facility,” said Jason Richey, president of LivaNova’s U.S. Region and general manager of the Neuromodulation Business Franchise. “This treatment gap means over one million individuals in the U.S. with undertreated epilepsy remain at risk for dangerous side effects of continued seizures, including injury, depression, memory issues and death.”
To view the multimedia release go to:
http://www.multivu.com/players/English/7966451-livanova-vns-therapy-epilepsy/
Categories //
Miscellaneous
Added:
3157 days ago by
MultiVuVideos
Runtime: 5m11s | Views: 1092 |
Comments: 0
Not yet rated |
|